video
Key Points For Success In A Clinical Trial
Interview featuring Frontage's Clinical Service's VP, Gary Steidl to discuss key points for success in a clinical trial.
Register to gain access to gated resources.
"*" indicates required fields
You must register to view this resource.
Resources to Consider


ebook
Alignment on Laboratory Support for Clinical Trials
In this Q&A, Nan Zhang MD PhD, Vice President of Global Central Lab and Biomarker Services...
poster
Utilization of High Sensitivity Proteomics NULISA HT Panels for...
This first-ever reported study demonstrates the rigorous qualification of NULISAseq high-t...

poster
Nonclinical Vehicle Formulations:HP-β-CD...
Beta-cyclodextrin is a commonly used excipient in nonclinical vehicle formulations. In a r...

white-paper
Accurate Mass: The best solution for metabolite identification in...
In this paper, we review appropriate definitions and describe the theoretical basis of why...

journal
An Ultrasensitive LC-APPI-MS/MS Method for Simultaneous Determination...
An Ultrasensitive LC-APPI-MS/MS Method for Simultaneous Determination of Ciclesonide and A...
case-study
Clinical Study of Abuse-Deterrent Formulation to Suppress Release of...
A number of the abuse-deterrent formulations in development and available on the market ut...

white-paper
Recruiting Women for First-in-Human Trials: Tips for Success
Programs that need to include women in their FIH (first in human) studies will find that s...

poster
EBF 2025: A generic immunogenicity assay for the detection of...
Discover how Frontage developed and qualified a generic ECLIA bridging immunogenicity assa...
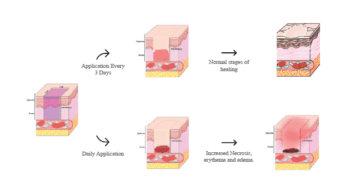
poster
ACT 2025 Study Design Considerations for Wound Healing Research in...
Discover how Frontage’s Safety & Toxicology team optimized wound healing study design in...

poster
AAPS 2025: Establishing Linear Viscoelastic Region and Gelation...
By defining the linear viscoelastic region and gelation dynamics, this work provides key i...

Duplex Sequencing: A Powerful Tool for In Vivo Gene Mutation
Discover our EMGS 2025 poster showcasing how Duplex Sequencing (ecNGS) reveals the genotox...



