white-paper
Recruiting Women for First-in-Human Trials: Tips for Success
Programs that need to include women in their FIH (first in human) studies will find that successful enrollment of women is achievable; however, it requires a concerted effort, with the correct strategies and proper support. In this paper, we explore the challenges in recruiting women for FIH studies and present a set of practices that have proven successful in improving the recruitment rates of women for these studies.
Register to gain access to gated resources.
"*" indicates required fields
Resources to Consider


infographic
6 Reasons To Do Exploratory Toxicology for Successful GLP Studies
Performing robust exploratory tests upfront can save time and money later on in the proces...

video
Key Points For Success In A Clinical Trial
Interview featuring Frontage's Clinical Service's VP, Gary Steidl to discuss key points fo...
poster
Utilization of High Sensitivity Proteomics NULISA HT Panels for...
This first-ever reported study demonstrates the rigorous qualification of NULISAseq high-t...

poster
EBF 2025: A generic immunogenicity assay for the detection of...
Discover how Frontage developed and qualified a generic ECLIA bridging immunogenicity assa...
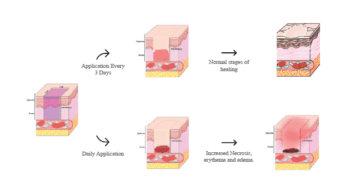
poster
ACT 2025 Study Design Considerations for Wound Healing Research in...
Discover how Frontage’s Safety & Toxicology team optimized wound healing study design in...

poster
AAPS 2025: Establishing Linear Viscoelastic Region and Gelation...
By defining the linear viscoelastic region and gelation dynamics, this work provides key i...

Duplex Sequencing: A Powerful Tool for In Vivo Gene Mutation
Discover our EMGS 2025 poster showcasing how Duplex Sequencing (ecNGS) reveals the genotox...
poster
AAIC 2025: Comparison of Plasma pTau217 Assays on Different Platforms
Plasma pTau217 was recently discovered as a prominent blood-based biomarker in the early d...

poster
WRIB 2024: High-quality RNA-seq Library Preparation from...
Here, we standardized the RNA extraction method from FFPE slides and an RNA-seq operation ...