webinar
General Practices for Critical Reagents Usage in Bioanalytical Labs
In this webinar, Dr. Santosh Shah discusses the general industry standards for utilizing critical reagents in bioanalytical labs, importance of bridging various lots of critical reagents and maintaining stocks, and reviews proper storage, management, and documentation of critical reagents.
Register to gain access to gated resources.
"*" indicates required fields

Resources to Consider


webinar
Best Practices in Managing IND Programs with CROs
As biotech and pharma companies have become more streamlined and “virtual”, the use of...
panel-discussion
Utilizing Ligand Binding Assays (LBA) and Critical Reagents for Drug...
Critical reagents – the key components used in LBA – are central to ensuring the consi...
poster
Utilization of High Sensitivity Proteomics NULISA HT Panels for...
This first-ever reported study demonstrates the rigorous qualification of NULISAseq high-t...

poster
EBF 2025: A generic immunogenicity assay for the detection of...
Discover how Frontage developed and qualified a generic ECLIA bridging immunogenicity assa...
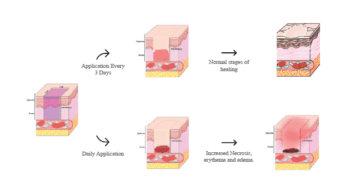
poster
ACT 2025 Study Design Considerations for Wound Healing Research in...
Discover how Frontage’s Safety & Toxicology team optimized wound healing study design in...

poster
AAPS 2025: Establishing Linear Viscoelastic Region and Gelation...
By defining the linear viscoelastic region and gelation dynamics, this work provides key i...

Duplex Sequencing: A Powerful Tool for In Vivo Gene Mutation
Discover our EMGS 2025 poster showcasing how Duplex Sequencing (ecNGS) reveals the genotox...
poster
AAIC 2025: Comparison of Plasma pTau217 Assays on Different Platforms
Plasma pTau217 was recently discovered as a prominent blood-based biomarker in the early d...

poster
WRIB 2024: High-quality RNA-seq Library Preparation from...
Here, we standardized the RNA extraction method from FFPE slides and an RNA-seq operation ...