Discovery
Medicinal Chemistry Services
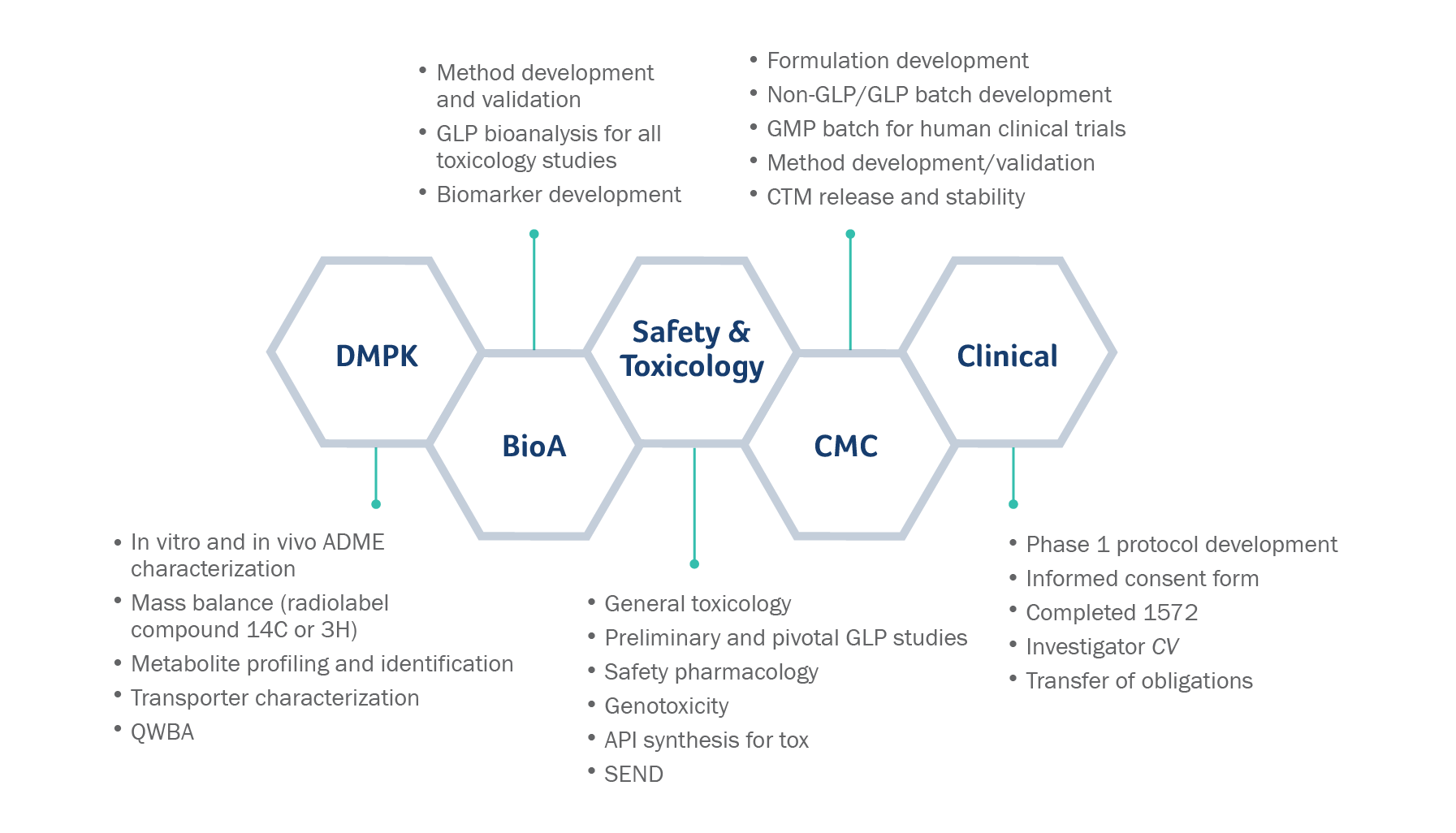
Discovery ADME
- Metabolic Stability
- CYP Interaction
- Permeability & Transporters
- Physicochemical Properties
- Protein Binding
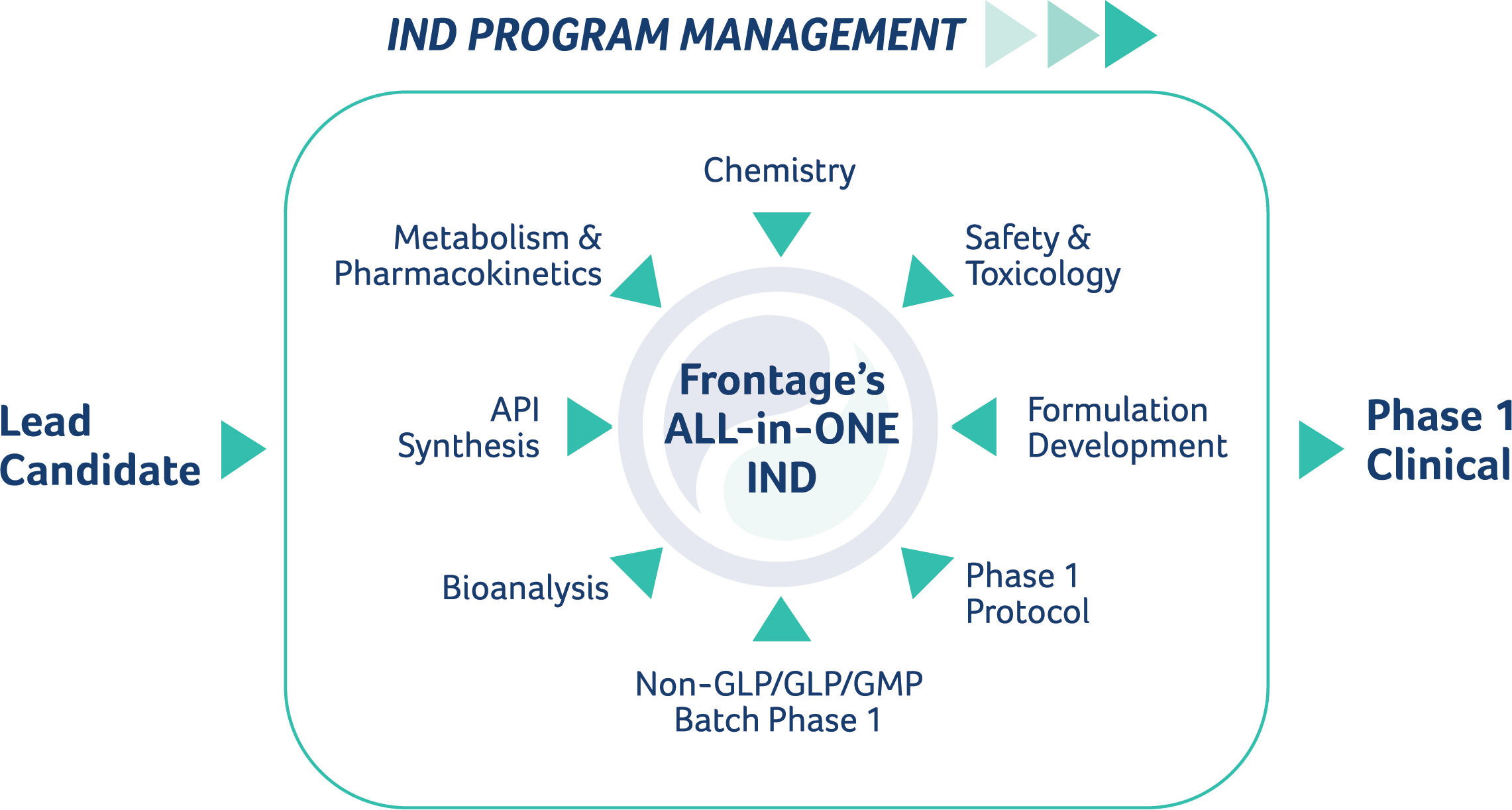
Managing an Investigational New Drug (IND) program and submission is a daunting task, requiring extensive planning and ongoing coordination of multiple scientific disciplines, each interrelated and reliant upon the other to achieve each successive milestone. At Frontage, we simplify the effort and reduce the risk by offering all of the critical IND-enabling services required to bring your lead candidate to Phase 1 in an organization sized to minimize complexity and maximize communication – ALL-in-ONE.
Whether you are a small start-up or a larger pharmaceutical client, we have the expertise and capability to customize a program to move your IND program from start to finish.
At Frontage, we offer a complete range of services to move from lead candidate selection to first-in-human. By providing the right comprehensive end-to-end services, we can accelerate your IND program. Consolidating your studies with Frontage provides the benefit of consistent quality, enhanced efficiency, and optimal cross-functional communications, ALL-in-ONE.
Have IND-enabling studies to run? Request a meeting.
At Frontage, we believe that effective communication underlies the success of everything we do, and as a mid-size CRO, we are the right size to ensure we are always focused on our customers and their needs. Timely, consistent, and clear communication, both internally and with our customers, is essential to ensuring successful outcomes.
We recognize that every client has unique and specific needs. The Frontage team of qualified project managers, study directors, and scientists work on a coordinated basis to listen to you and to make sure that your expectations are clearly understood.
Our ALL-in-ONE suite of IND enabling services, coupled with our right-size organization, ensures nimbleness of response that can take months off a typical IND timeline. That is what having the right services and being the right size is all about.
Regardless of your program size, our dedicated project leaders will work closely with your team to ensure your project gets the attention to detail and support it deserves.