Safety Assessment & Toxicology
A full spectrum of Safety & Toxicology services from IND enabling to NDA submission.
Download Brochure
Streamlining your development process
Operating at sites in the US, Canada and China, Frontage provides all the resources you need to support your nonclinical safety research and development plans.
Our team of highly-experienced Study Directors, technicians, and program managers will help guide you through the critical phases of toxicity testing, from pilot tolerability and dose range-finding studies to definitive GLP toxicology studies required for the support of human safety.

Safety Pharmacology
GLP Regulatory and Non-GLP Discovery Pharmacology Testing

Genetic Toxicology
Essential for assessment of genotoxic hazards

Toxicology
Moving client compounds swiftly into human studies
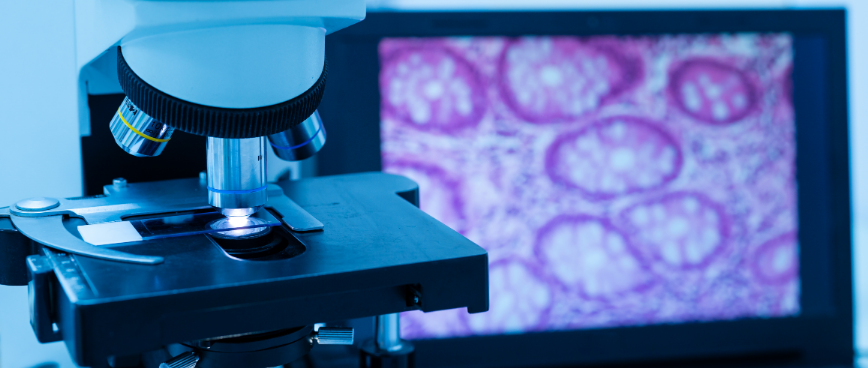
Global Pathology Services
Comprehensive characterization of tissue samples to assure accurate safety assessment

Toxicokinetics
TK modeling in support of IND-enabling GLP studies

Standard for Exchange of Nonclinical Data (SEND)
SEND datasets for toxicology studies
Pilot / Non-GLP Studies
The early phase screening and pilot tolerability/PK studies conducted in both rodent and nonrodent models form the foundation for the subsequent definitive (GLP) toxicology studies and are important elements in a successful, nonclinical safety evaluation program. These studies include maximum tolerated dose (MTD), repeat-dose range-finding, exploratory PK, and feasibility studies needed to justify the design and dose-level considerations for the pivotal studies to follow.
GLP Studies
Frontage GLP toxicology studies have supported hundreds of successful client regulatory submissions globally. All standard dosing regimens and species (including nonhuman primates) are supported and are conducted in compliance with international GLP and animal welfare regulations.
Preclinical Safety: IND-Enabling and Later-Phase Toxicology Studies
Our GLP studies are conducted in a wide range of animal models as appropriate for either small molecule therapeutics or biologics, across a wide variety of therapeutic indications. These studies are often part of IND-enabling programs as well as later-phase development chronic, reproductive/developmental, and carcinogenicity studies needed to support large-scale human trials and marketing permits.
Our fully-integrated IND-enabling programs include general toxicology plus genetic toxicology and safety pharmacology as appropriate, plus all affiliated analytical / bioanalytical support elements, enabling the collection of robust and comprehensive data packages for IND submission and support. From pilot screening to IND-enabling, and ultimately to studies needed for the NDA, we offer comprehensive expertise to help design, execute, and report the studies required to satisfy your planning and regulatory safety obligations.
Check Out Our Publication in International Journal of Toxicology
Hepatitis A Virus Infection in Cynomolgus Monkeys Confounds the Safety Evaluation of a Drug Candidate
Download the articleResources To Consider

Safety and Toxicology Services Brochure
Guidelines for Developmental and Reproductive Toxicity (DART) Testing

IND Enabling Services Brochure


