Central Lab Services
A Next Generation Central Lab to Accelerate your Clinical Trial and Elevate Patient Care.
Learn More
Frontage Laboratories’ Central Laboratory Services deliver end-to-end global support for clinical trials from early to late phases, combining scientific excellence with fully integrated logistics.
Our CAP-accredited and CLIA-certified facilities provide a broad menu of safety, efficacy, and biomarker testing across hematology, chemistry, immunology, genomics, and specialized assays. Beyond testing, Frontage offers comprehensive operational solutions including clinical kit design and production, global kit and specimen shipment management with temperature-controlled logistics, laboratory manual development and maintenance, specimen reconciliation and archival tracking, as well as Site Initiation Visit (SIV) and sample management training for investigator sites. With harmonized operations across the U.S., EU, and China, supported by robust data management systems and a dedicated logistics team, Frontage ensures accurate, timely, and compliant results that help sponsors streamline global studies and meet regulatory expectations worldwide.

A Comprehensive Suite of Services
At Frontage, a thorough approach ensures complete alignment of laboratory support services to meet the ever-changing requirements of clinical trials.
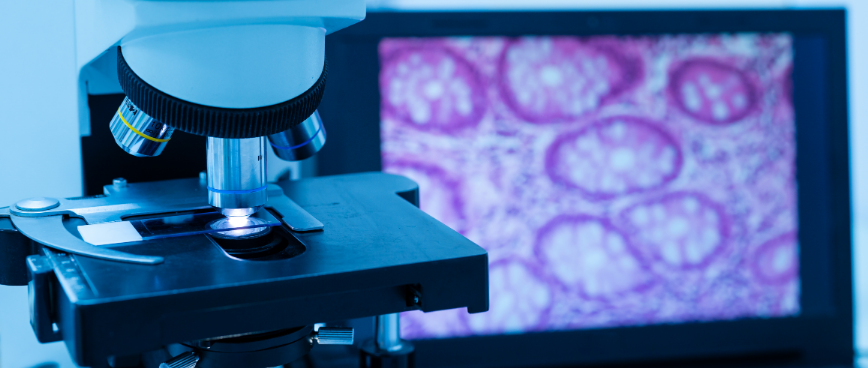
Histology & Pathology
Expertise in sample processing with a wide range of stains including immunochemistry and multiplexing IHC.
Safety Testing & Specialty Lab Testing

Biorepository Services
Our Biorepository is a fundamental key component of Central Labs comprehensive suite of services

Clinical Trial Logistics Services
Frontage has years of experience providing excellent project management support & logistics services.

Clinical Sample Processing
Clinical sample processing services

Biospecimen Data Services
Comprehensive Data Solutions for Clinical Trials

Central Lab e-Portal

Sample Management
Optimize Your Clinical Trials with Streamlined Sample Management

Central Lab Clinical Trial Request Intake Form
What makes us different?
- Integration: We are aligned with other laboratory testing services such as bioanalytical and biomarker services.
- Sample Management System: A Dedicated Biorepository Laboratory Information System ensures that samples are received, sorted, and logged in based on the barcode, sent to the lab for immediate testing, and tracked throughout the entire process; Long-term storage at 4°C, -20°C, -70°C, and liquid nitrogen storage in a secure sample management room is available through Frontage Biorepository Services. Samples can also be transferred to auxiliary labs (including packaging and re-shipping), returned, or disposed of.
- Data Consistency: Consistent methodologies across our global central lab services locations to ensure laboratory data consistency globally and over time.
- State of the Art Instrumentation and Laboratory Information System: Accurate and precise laboratory results, as well as capability in handling high testing volume per day. Cloud-based LIS technology with emphasis on sample management, logistics, data regulatory compliance, and data analysis. Data mining based on Artificial Intelligence is an option.
- Responsiveness: The Frontage central laboratory will design each job and role to support operational agility in line with industry best practices.
- Team-Based Approach for Project Management: This approach provides seamless communications and continuity of projects to ensure the utmost client satisfaction.
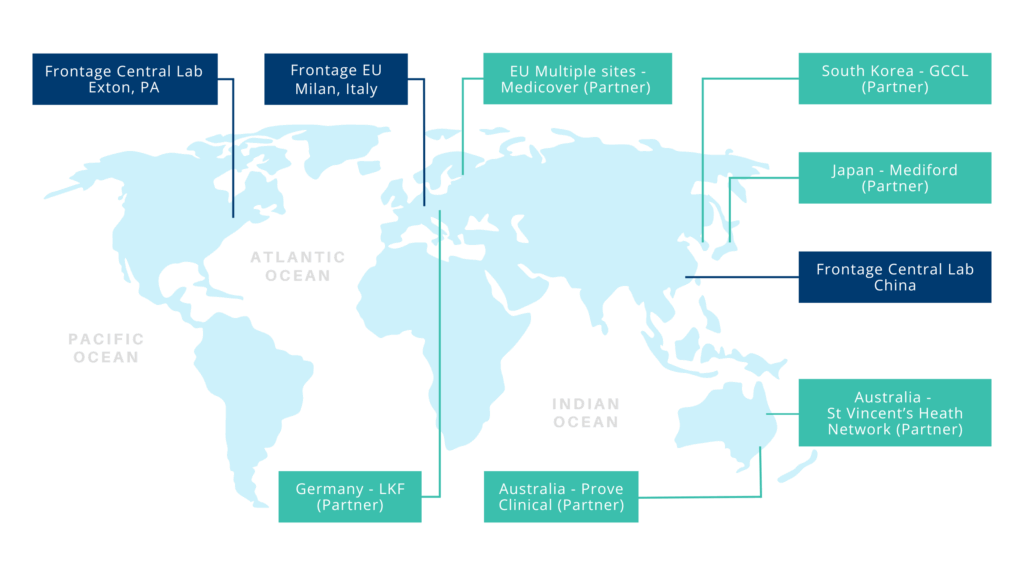
Global Coverage
From North America to Europe and Asia Pacific, our global team provides comprehensive support services for pharmaceutical and biotechnological companies across the world.
Next Generation Central Lab
Watch our webinar outlining the next general central lab services that provide lab assessments, kits, logistics support, and delivery of data in certain formats.
Download our webinar
