Resource Library
/
Meeting public health and regulatory demands for next generation consumer products
Resource Library
/
Meeting public health and regulatory demands for next generation consumer products
panel-discussion
Meeting public health and regulatory demands for next generation consumer products
Register to gain access to gated resources.
"*" indicates required fields

Resources to Consider

white-paper
Evaluating Emerging Tobacco Products and Their Potential Impact on...
Decades of studies have conclusively demonstrated that smoking combustible cigarettes, as ...

webinar
Webinar Analytical Challenges for CMC Development of Cell and Gene...
In this webinar we will cover:
Innovative solutions and best practices for accelera...
Innovative solutions and best practices for accelera...

webinar
Microbiological Testing Considerations for Pharmaceutical Products
In this webinar, speaker Christopher Gilmer introduces microbiological testing for raw mat...
poster
Utilization of High Sensitivity Proteomics NULISA HT Panels for...
This first-ever reported study demonstrates the rigorous qualification of NULISAseq high-t...

poster
EBF 2025: A generic immunogenicity assay for the detection of...
Discover how Frontage developed and qualified a generic ECLIA bridging immunogenicity assa...
poster
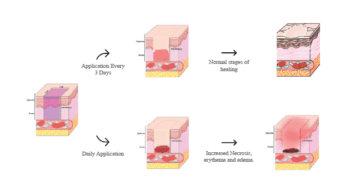
ACT 2025 Study Design Considerations for Wound Healing Research in...
Discover how Frontage’s Safety & Toxicology team optimized wound healing study design in...

poster
AAPS 2025: Establishing Linear Viscoelastic Region and Gelation...
By defining the linear viscoelastic region and gelation dynamics, this work provides key i...

Duplex Sequencing: A Powerful Tool for In Vivo Gene Mutation
Discover our EMGS 2025 poster showcasing how Duplex Sequencing (ecNGS) reveals the genotox...
poster
AAIC 2025: Comparison of Plasma pTau217 Assays on Different Platforms
Plasma pTau217 was recently discovered as a prominent blood-based biomarker in the early d...