brochure
Frontage Laboratories Company Overview Brochure
Frontage Laboratories, Inc. is a full-service CRO providing integrated, scientifically-driven research, analytical and development services. With over 20 years of experience, we have successfully assisted our clients in advancing hundreds of compounds through the drug development process.
Register to gain access to gated resources.
"*" indicates required fields
Resources to Consider


virtual-tour
Frontage Hayward, CA Site Virtual Tour
Frontage’s newest facility in Hayward, CA started operations in 2022. With over 19,000 i...

virtual-tour
Frontage 760 Site Virtual Tour
Frontage Virtual Tour: Do a 3D walkthrough of our site at 760 Pennsylvania Dr, Exton, PA 1...

white-paper
IVRT Insights From Frontage CMC Experts
In this white paper, we detail IVRT methods pertaining to generic and innovator products a...
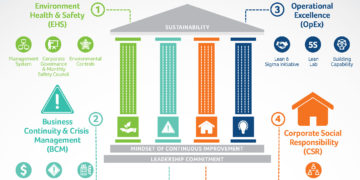
infographic
Sustainability Initiatives at Frontage
Consistent with our company’s vision, mission, and values, we are committed to pursuing ...

virtual-tour
Frontage Holdings Launched A New 42,000 Sq.Ft. Lab In Shanghai, China
Frontage Holdings Corporation (“Frontage Holdings” or “Frontage,” stock code: 1521...

video
Dr. Song Li, Frontage’s Founder, Ringing The Bell Of The Hong Kong...
Frontage Holdings Corporation, the parent company of Frontage Laboratories, Inc., a fast-g...

fact-sheet
Overview of Cell-Free Assay Services
Throughout years, our experts have developed comprehensive screening assays for 350+ targe...

fact-sheet
Global Drug Discovery Services Overview
Frontage’s discovery team is comprised of experienced medical chemists, biologists,and D...

fact-sheet
Clinical Services Overview
Frontage provides comprehensive clinical trial support to ensure rapid start-up and execut...

fact-sheet
Bioanalytical Services Overview
Frontage Laboratories provides bioanalytical services to help support your full drug devel...
brochure
Safety and Toxicology Services Brochure
Frontage offers an impressive suite of services to take lead candidate drugs to the IND st...

brochure
IND Enabling Services Brochure
At Frontage, we simplify the effort of Investigational New Drug (IND) and reduce the risk ...


