 Resource Library
/
Characterization of Positional Isomers of Interchain Cysteine Linked Antibody−Drug Conjugates by High-Resolution Mass Spectrometry
Resource Library
/
Characterization of Positional Isomers of Interchain Cysteine Linked Antibody−Drug Conjugates by High-Resolution Mass Spectrometry
journal
Characterization of Positional Isomers of Interchain Cysteine Linked Antibody−Drug Conjugates by High-Resolution Mass Spectrometry
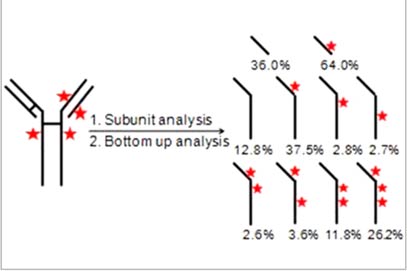
Interchain cysteine linked antibody–drug conjugates (ADCs) are emerging therapeutic products that antagonize cancers. The toxic payloads are selectively linked to the interchain cysteines and generate heterogeneous mixtures of positional isomers. These positional isomers might contribute differently to the therapeutic efficacy because of the variation in conjugation stability, and thus they need to be well characterized. However, the characterization of the positional isomers of interchain cysteine linked ADCs is very challenging, mainly because of the high similarity between those isomers. In this research, we developed a novel mass spectrometry method for the characterization of positional isomers of interchain cysteine linked ADCs. The subunit analysis and the bottom-up analysis provided abundant information about the drug numbers and drug linking positions on each chain. Because the method can provide accurate data on drug linking numbers and positions on each chain, it will be very useful for researchers in cancer drug development and cancer treatment.
Register to gain access to gated resources.
"*" indicates required fields
Resources to Consider


Accurate determination of drug-to-antibody ratio of interchain...

Characterization of Interchain Cysteine Linked Antibody Drug...
Bioanalysis of antibody-drug conjugates (ADCs) by LC/MS: challenges...

Bioanalytical Strategies for PK Analysis of Antibody-Drug Conjugates...

Determination of Pregabalinin Human Plasma by Liquid...

Quantitative Assessment of mRNA Drug Biological Activity by...

Quantitative Analysis of Oligonucleotide by Ion Paring Reversed-Phase...

IN Vitro Assessment of the inhibition of Human Organic Anion...

A Hybrid Trial Approach by Combining 13C8-Lableded & Radiolabeled...

Quantitative Analysis of Antibody Conjugate Payload by LC MS/MS...

Achieving sensitive and specific oligonucleotide quantification by...

