Resource Library
/
BSEPcyte® and MDR3cyte®: Innovative Solutions for Investigating Drug-Induced Liver Injury
Resource Library
/
BSEPcyte® and MDR3cyte®: Innovative Solutions for Investigating Drug-Induced Liver Injury
webinar
BSEPcyte® and MDR3cyte®: Innovative Solutions for Investigating Drug-Induced Liver Injury
Drug-induced liver injury (DILI) accounts for >50% acute liver failures, and is the leading cause of drug development failure, boxed warning and market withdrawal of approved drugs. Inhibition of BSEP and MDR3 is one of the underlying mechanisms for DILI. BSEP and MDR3 are the primary hepatic transporters responsible for exporting bile salts and phosphatidylcholine, respectively. In humans, inhibition of BSEP and/or MDR3 can result in serious liver injury. This presentation will discuss our patented platforms BSEPcyte® and MDR3cyte® using primary hepatocytes in suspension for higher throughput assessment of DILI potentials. In addition, a brief review of the critical transporter studies to inform DDI potentials will also be discussed. This presentation will benefit scientists wanting to learn more about DILI and/or those needing guidance on crucial transporter studies for better assessment of DDI potentials that is aligned with regulatory guidance.
Register to gain access to gated resources.
"*" indicates required fields

Resources to Consider


Strategies and Solutions for Early Phase Formulation Development and...

Challenges and Bioanalytical Solutions of Ultra-Low Sensitive 4-in-1...

CMC-Product Development & Manufacturing Integrated Solutions
Bioanalysis of antibody-drug conjugates (ADCs) by LC/MS: challenges...

Development of Stable Nanoemulsion Formulation as an Ophthalmic Drug...
Utilization of High Sensitivity Proteomics NULISA HT Panels for...

EBF 2025: A generic immunogenicity assay for the detection of...
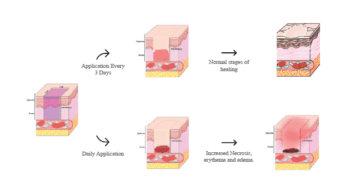
ACT 2025 Study Design Considerations for Wound Healing Research in...

AAPS 2025: Establishing Linear Viscoelastic Region and Gelation...

Duplex Sequencing: A Powerful Tool for In Vivo Gene Mutation
AAIC 2025: Comparison of Plasma pTau217 Assays on Different Platforms