Frontage Clinical Services, Inc. Expands Facilities to Accommodate Tobacco and Nicotine-Related Studies
Frontage Clinical Services, Inc. (“Frontage Clinical”), an associate of Frontage Laboratories, Inc. (“Frontage Labs”), is pleased to announce completion of renovations to Frontage Clinical’s center in Secaucus, New Jersey, to now accommodate clinical studies on tobacco and nicotine-containing products. This expansion highlights Frontage Clinical’s commitment to the investigation of the safety of new tobacco alternatives.
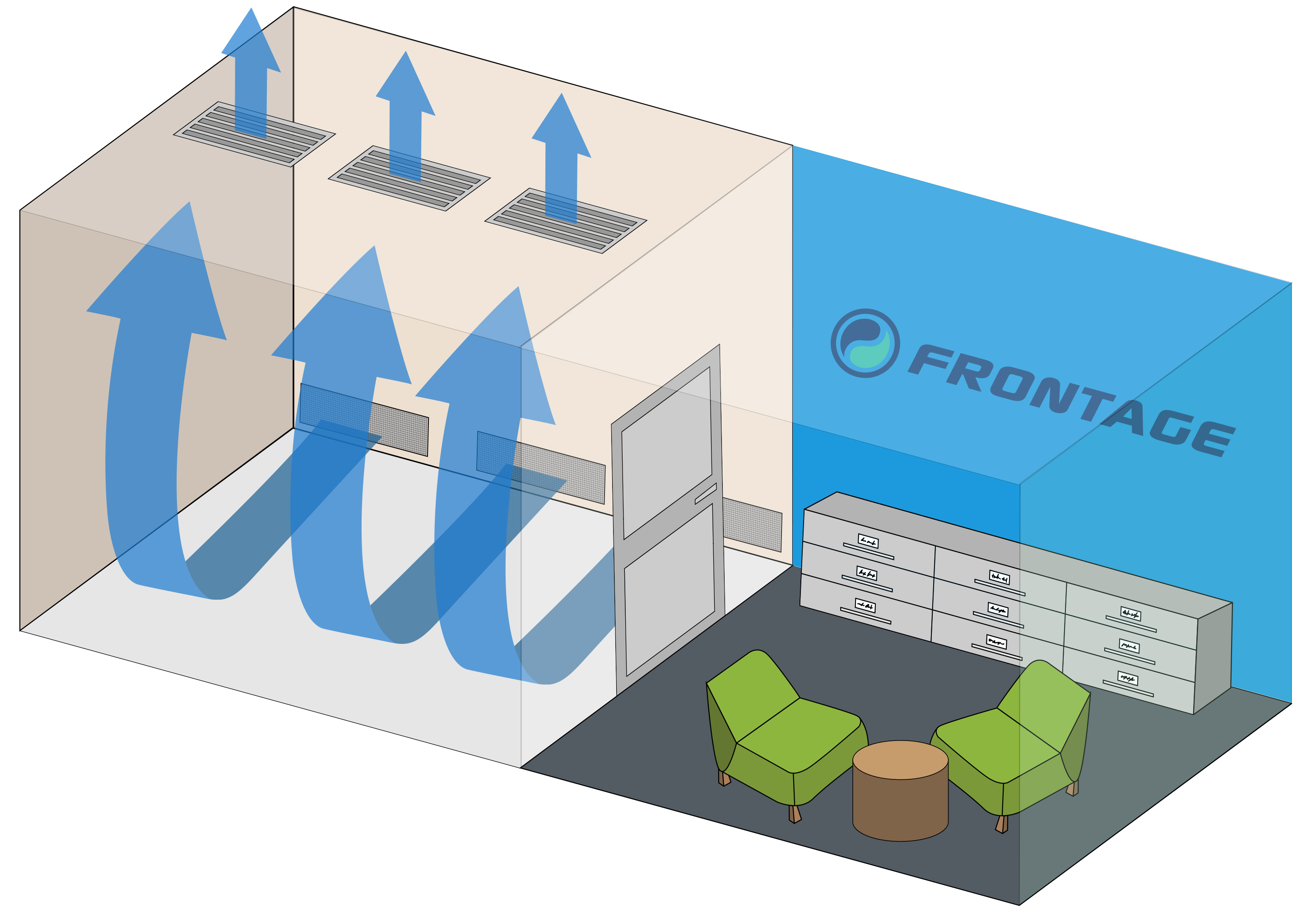
The addition of two dedicated negative-pressure rooms that utilize displacement ventilation enables evaluation of subjects using a variety of investigational tobacco products, including combustible cigarettes and electronic nicotine delivery systems (ENDS), as well as smoking cessation therapies.
“Incorporation of these specialized clinical facilities enables us to perform clinical studies to assess product use behavior, abuse liability, product risk assessment and pharmacokinetic analyses. Frontage Clinical is committed to providing the best services possible for each of its partners. The experienced clinical staff, PIs and scientists are eager to meet the ever-evolving needs of this industry.” Said by Lisa Diamond, Ph.D., President of Frontage Clinical.
To complement Frontage Clinical’s operations expansion, Frontage Labs’ bioanalytical lab is developing a portfolio of assays to evaluate tobacco-specific biomarkers of exposure and potential harm. This allows Frontage to offer integrated services to their clients to provide high-quality data to meet regulatory submission requirements.
“Our bioanalytical facility is top-notch with an experienced team, specialized laboratory equipment, and dedicated space to perform these challenging biomarker assays.” John Lin, Ph.D., Executive Vice President, Bioanalytical and Biologics Services of Frontage Labs.
In support of the mandated FDA public health initiative, Frontage Clinical and Frontage Labs are committed to assisting their clients in evaluation of the risks and safety profiles of investigational tobacco products. The addition of these capabilities positions Frontage Clinical and Frontage Labs to offer a meaningful contribution towards the promotion of public health.
About Frontage Clinical Services, Inc.:
With over 20 years’ experience in the execution of comprehensive Phase I-IIa studies, the Frontage Clinical team collaborates closely with our clients to develop a unique approach to overcome the challenges of each study, while maximizing efficiency and mitigating risk. We leverage our best practices in study design and execution to advance your investigative compound to a drug candidate. Located near NYC, we have access to one of the most diverse study populations and acclaimed medical professionals and facilities to help support your study.
About Frontage Laboratories, Inc.:
Frontage Laboratories, Inc. is a contract research organization (CRO) that provides integrated, science-driven, product development services throughout the drug discovery and development process to enable pharmaceutical and biotechnology companies to achieve their development goals. Comprehensive services include drug metabolism and pharmacokinetics, analytical testing and formulation development, preclinical and clinical trial material manufacturing, bioanalysis, preclinical safety and toxicology assessment and early phase clinical studies. Frontage has enabled many biotechnology companies and leading pharmaceutical companies of all sizes to advance hundreds of molecules through development and file regulatory submissions in the United States and China.

