Current Clinical Research Studies
Call now, 1-877-298-9071 (press option 1 for recruitment) to see if you prequalify to come in for screening. Refer a friend who participates and receive a referral bonus.

Y-4-LC-02 (US) Phase 1 Study
Participate in a Phase I Clinical Trial
Phase 1 study designed to compare how a new investigational medication (Y-4) behaves in the body versus its individual components, pregabalin and riluzole, when taken on an empty stomach. The study will also assess the safety and tolerability of these medications in healthy adults. Compensation up to $9,050 for your time and participation.
To participate, you must:
- Be a Healthy Adult
- Be 18 to 45 years old
- Have a BMI between 19 to 28 kg/m2
- Be a Non-Smokers, Non-Vapers and not used other Tabacco Products
- Have a Good Venous Access
Study Requirements:
- Period 1 (Days 1–4): Y-4 tablet (combination of pregabalin and riluzole) with monitoring for 3 days
- Period 2 (Days 8–11): Pregabalin capsule with monitoring for 3 days
- Period 3 (Days 15–18): Riluzole tablet with monitoring for 3 days
- Discharge on Day 18 followed by 1 follow-up phone call about 7 days later
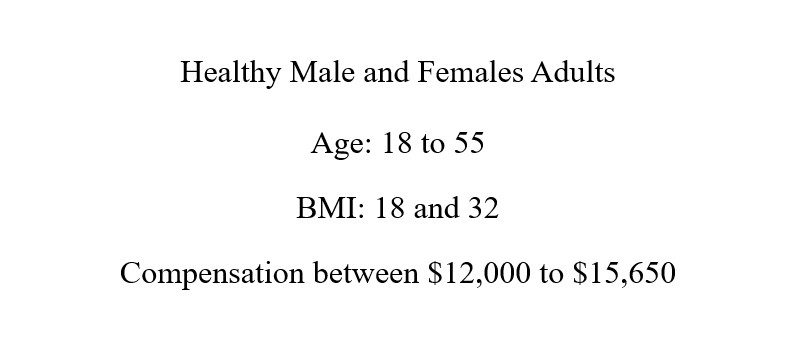
HNC364 – Phase I Clinical Trial
Participate in a Phase I Clinical Trial
- Compensation between $12,000 to $15,250 for your time and participation.
This is a Phase 1 research study to compare a investigational injectable medication (HNC364) with an approved oral medication called AZILECT. The goal is to see how the new drug works in the body, and to check its safety and how well it is tolerated.
Eligibility Criteria
- Be a Healthy Adult
- Be 18 to 55 years old
- Have a BMI between 18 to 32.0 kg/m2
- Be a Non-Smokers, Non-Vapers and not used other Tabacco Products
- Have a Good Venous Access
Study Requirements:
If you are chosen for the investigational Injection Group (HNC364):
- You will receive 4 study drug injections in the shoulder, once every 4 weeks.
- Total study length: about 5–6 months
- Clinic stay: 4 overnight stays (one per dose)
- Additional 4 outpatient visits between doses
- Requirement: 4 separate one night stay in the clinic and 4 follow-up visits in between doses
- Compensation: up to $12,000 upon completion
If you are chosen for the Tablet Group (AZILECT):
- You will take a pill once daily for 25 days
- Total study length: about 2 months
- Clinic stay: about 25 consecutive overnights
- 1 follow-up visit after discharge
- Requirement: 26 overnight stays (27days) and one follow-up visit
- Compensation: up to $15,250 upon completion

New Asian Volunteers Needed
✨ Frontage Clinical Services is Looking for Asian Volunteers! ✨
Be part of our upcoming clinical studies and help advance medical science.
Why Join?
✔ Contribute to important medical research
✔ Support the development of future treatments
✔ Play a key role in improving healthcare for all communities
📢 We welcome volunteers from all backgrounds to join our clinical research community. We’re especially interested in increasing participation from Asian communities to help make research more inclusive and representative.

Researchers are studying a new investigational nasal spray version of semaglutide.
Participate in a Phase I Clinical Trial
- Compensation is $4,225 for your time and travel.
Eligibility Criteria
To participate, :
- You must be a Healthy Adult
- You must be 18 to 65 years old
- You must have a BMI between 27 to 38kg/m2
- You must have had stable weight (no major weight change) over the last 3 months
- You can smoke more than 5 but no more than 10 cigarettes per day and be willing to refrain from smoking for the entire duration of the study.
Study Requirements:
- Healthy adult ages 18–65 with overweight or obesity may qualify. One screening visit, 4-night clinic stay plus 9 outpatient visits. Compensation $4,225.
Surgically Sterile Participants
- Males who have undergone a vasectomy
- Females who have had a surgical sterilization procedure such as tubal ligation, hysterectomy, oophorectomy.

New Volunteers!
- New to the clinic
- Paid Compensation
- Healthy Male and Female participants
- Must be 18 years old and older
- Willing to participate in overnight studies

Referral Program
Frontage Clinical Services is looking for volunteers to participate in clinical research studies!
Be compensated for referring a friend!
Ask us about our Referral Program
How does it work?
- Refer a friend/family member who has never
contacted Frontage Clinical Services before to call
us about participating in one of our research
studies. - Your referral must give your name and telephone
number to our recruitment staff during the initiation of the profile creation! - If your friend/family member completes a study
with us, you will receive a bonus for each referral. - It’s that simple!





